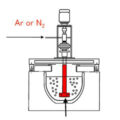
On-line molten metal degassing is usually accomplished by blowing an inert gas containing a certain percentage of chlorine (Cl2) gas. Chlorine forms small bubbles in molten metal. Degassing is usually carried out in a continuous operation before casting, which itself can be carried out continuously. A mixture of inert gas and Cl2 (chlorine) is injected into the molten metal to treat the molten metal as it flows from the furnace to the pouring pit. Although only inert gas can effectively remove dissolved H2 (hydrogen) through mass transfer, it is necessary to remove alkali metal and alkaline earth metal impurities (such as sodium (Na), lithium (Li) and calcium (Ca)) in molten metal. Reactant, such as Cl2.

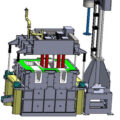
In order to achieve effective degassing, all degassing equipment must deliver a certain amount of minimum gas per kilogram of metal. The molten metal can be degassed online in a trough or deep box degasser. In the trough degasser, the residence time of the metal in the gas-providing area is much shorter than in the deep box degasser. Each rotary ejector must deliver a large amount of gas and has a suitable delivery capacity. The amount of gas determines the effectiveness of the ejector design.
It has been noticed that in a trough degasser with a gas rotor, the gas rotor can deliver an appropriate amount of gas to the molten metal, and the gas tends to be released from the rotor in an irregular manner, resulting in molten metal splashing on the surface and low efficiency. Some trough degassers use several relatively small rotary gas ejectors along the length of the trough section to achieve the effect of a continuous or pseudo “plug” flow reactor, rather than a fully mixed flow reactor or a continuously stirred tank The effect of the type reactor, this is a deep box deaerator. In an ideal plug flow reactor, there is no mixing, and the fluid elements leave in the same order they arrive.
However, a trough-shaped degasser with multiple small rotating gas ejectors cannot deliver a large amount of gas in the form of fine bubbles into molten metal without obvious gas flow irregularities, so it is not suitable for applications requiring fine bubbles. The form is large-scale natural gas transportation. When using chlorine gas as a degassing agent, the deep box degasser removes hydrogen and inclusions from the molten metal more effectively than the trough degasser. When a salt flux is used instead of chlorine, the same result can be expected. Therefore, a deep box type degasser must be used to reduce splashing on the surface of molten metal and maximize the removal efficiency of dissolved gases and inclusions.